The “Three R principles” are quite old but represent a “must-have” (or rather a “must-be”) in our daily lab routine. In 1954, the Universities Federation of Animal Welfare (UFAW) approached two scientists who were instructed to inaugurate a systematic study of laboratory techniques in their ethical aspects. Based on their reports, a book named “The 3Rs” was published in 1959, which describes guidelines for a more ethical use of animals in science (Russell and Burch, 1959). The 3Rs are: Replacement, Reduction, and Refinement. Replacement aims at generally avoiding or replacing animals in research. Reduction means to reduce the number of animals in science whilst obtaining the same amount of data or to obtain more data from the same number of animals. Refinement, however, exhorts to minimise potential pain, suffering, and distress and to enhance animal welfare for the animals used in science. Sounds like a book that should be lying on the nightstand of every scientist who works with animals.
Numbers and facts
In December 2016, the Federal Ministry of Food and Agriculture (German: Bundesministerium für Ernährung und Landwirtschaft, BMEL) published their annual report about the use of animals in research, in this case regarding the year 2015. Their data say that two years ago, around 2.8 million animals were used in German research laboratories of which most died for the sake of science. 71.16% of these animals were mice (just to give a number: this percentage correlates to 2,031,338 animals), 11.75% were rats, and 4.32% were zebra fish. 39.85% of the total used animals were genetically modified and most (81.5%) were used for basic science. In 2015, Bonn, in this regard, can be found on rank 21 of the currently total listed 91 cities in Germany, which are known to use animals in research (www.datenbank-tierversuche.de). Of note, this list is most likely beyond complete.
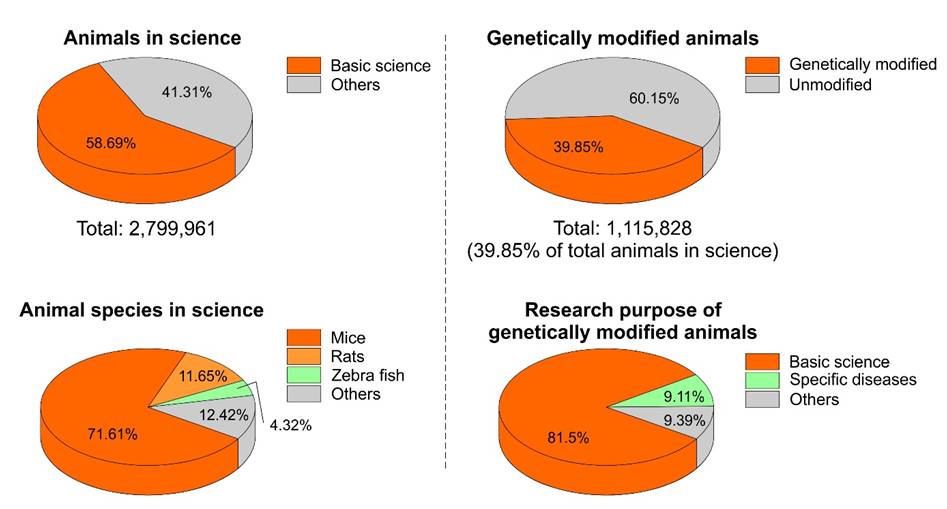
Figure 1: Number of animals used in German laboratories in 2015. Data were extracted from the annual report of the Federal Ministry of Food and Agriculture.
Are there alternatives?
Numbers like those mentioned above are eye-openers and everyone, especially people who actively contribute to animal-based research, should be aware of them. Public debates about the use of animals in research are omnipresent and refer to more and sometimes less approved information sources. Googling this topic can be day-filling and provides a long list of results, which is why the present article focuses on the question whether there are any (preferably good) alternatives to animal-based experiments available today. Of note, this article does not aim at discussing about the necessity of animal testing or the general use of animals in research. But, since the need for good alternatives to animal-based research in (basic) science is neither unfounded nor new, we searched for very obvious ones.
Being aware of the “sources” that are used in our daily lab routine has nothing to do with being a Peta activist or hugging trees in our free time. It only means that a person, in this case a scientist, really knows and understands his or her job. Replacing animals in (basic) science is not only the wish of the population group mentioned above, but actually also a major concern addressed by the BMEL and, thus, by the government. To reduce the number of animals used in research, the BMEL annually announces prize money of up to 25,000 € for researchers who push the development of methods that contribute to animal-free science. In 2015, this money was awarded to the working group around Prof Marcel Leist at the University of Konstanz, who developed a cell culture model based on neurons and astrocytes to study neurodegenerative diseases. So, here we go: The putative alternative to animal-based research could be… cell culture!

Cell culture can often replace studies in animals (image from colourbox.com/ motorolka)
Cell culture: curse and blessing
The culturing of e.g. eukaryotic cells is a widely used substitute for animal research in laboratories worldwide. Of note, the use of cell culture is also limited because cell lines cannot always completely mimic the in vivo situation. However, it displays a cost-efficient alternative to the expensive and work-intensive housing of animals and is connected to far less burdens than experiments that include animals. In cell culture, cells can be directly taken from an animal or human being (primary cells) and used for a variety of experiments. Since the reproducibility of these experiments sometimes is limping, the generation of immortalised and well-established cell lines has been perfected during the past decades. Here, cell lines like HeLa (cervical cancer cell line obtained from Henrietta Lacks) or CHO (Chinese Hamster Ovary cells) nowadays have been encountered by almost every student of life sciences. These immortalised cell lines are (more or less) easy to culture and, most importantly, have been used for experiments since the late 50s of the past century, which means that there is plenty of experience available. The initial source for these cells in culture still are animals (or humans), but the well-established ones from today all go back to one single animal, meaning that the Reduction aspect of the 3Rs has been implemented perfectly. It is well accepted amongst scientists that cell culture-based experiments provide reliable and well-reproducible results that are generated much faster than those obtained from animal-based studies. So far, so good. But, as usual, there is also a fly in this ointment.

On average one calf is needed to obtain one bottle of FBS (image from colourbox.com/ Paulo Vítor Martins)
The hushed-up reality
Cells in culture cannot exist from air and the researcher’s love alone. They need to be kept in a proper culture medium, which normally contains compounds such as amino acids, glutamine, and, most importantly, foetal bovine serum (FBS, also termed foetal calf serum or FCS). The latter is perplexing, because the supplementation with FBS means that commonly used cell culture media are not free of animal-based products. FBS is produced in many different countries and cannot be considered a waste product of the meat and leather industry anymore, since its need for research has increased during the past years. It was estimated that in 1995 about 500,000 litres of serum were produced on an annual basis (Hodgson et al. 1995). In 2008, the sales of FBS were estimated to be 700,000 litres worldwide. Considering the expansion of science during the past decade, this number has most likely increased drastically. On average, a calf foetus yields around 500 ml of serum, which means that at least one million foetuses were needed per year to only serve the need of cells in culture. General cell culture media contain around 10% FBS, which means that, considering a 500 ml medium bottle, the volume of 1/10th of a calf foetus has to be added to serve the need of an average cell population. Grossed up to the average use per year, that represents quite a herd of calves per lab.
Bovine foetuses for FBS production are obtained from pregnant cows after the dam’s neck has been cut. Depending on the animal welfare rules of the corresponding country, the blood from the still alive foetus is collected as early as 5 min or as long as 25-40 min after slaughter of the dam by puncturing the beating heart of the non-anesthetized foetus with large diameter needles (Jochems et al., 2002). Mammals like sheep are known to be conscious and aware of their surroundings once 70% of gestation has elapsed (Mellor and Gregory, 2003), which is why suggestions regarding the handling and killing of those unborn calves have been formulated during a workshop on “The humane collection of foetal bovine serum” in the Netherlands in 2003 (van der Valk et al. 2004). To ensure unawareness, the uterus of the dam is only cut half-open to hinder the calf from breathing, which, in combination with the immense blood loss of the dam, causes the calves’ death. This treatment is mandatory, since unconscious or dead animals cannot have noxious experiences and, therefore, cannot suffer. However, if these procedures to minimise pain and suffering of the animals are indeed applied, remains unknown. Using serum from adult cows would increase the yield per animal, but FBS is known to bear a very low gamma-globulin count, which is favourable for cell growth since high antibody contents may inhibit cell growth and proliferation.
Expanding old frontiers
Since the previous section might have caused some indisposition, it is worth to think about the pros and cons of FBS. The biggest (and almost invincible) pro of cell culture media containing FBS is very simple: they make cell culture work. Media containing FBS have been used since decades and the methodology here is well established. Most of the commercially available cell lines are recommended to be cultured in medium containing FBS and, normally, cells cultured in the appropriate, FBS-supplemented medium can be kept in culture for many passages without growth arrest. On the con side, however, there is also something going on: even at a time where green-fluorescent pigs are born, nobody really knows the ingredients of FBS. It contains several factors which are believed to stimulate the division and growth of cells, but, in the end, FBS remains an ambiguous factor. Since FBS is a natural product originating from different calves, properties and effects can differ from batch to batch and the serum is also prone to biological contaminations. And, as previously mentioned, ethics and animal welfare are reasonable issues that underline the need for a chemical alternative.
A common saying states “Never change a running system”. Still, it might be worth to learn more about possible alternatives. In 1993, the German newspaper “Der Spiegel” reported about the partially cruel incidents happening during the harvest of FBS and roughly 10 years later, the Swiss association “Fonds für versuchstierfreie Forschung”, in short FFvFF (English: Fund for animal-free research), approached Prof René Fischer, who, at that time, worked in the Hilvert lab at ETH Zürich. In 2001, Prof Fischer became head of a project called “Serumfrei” (English: serum-free), which aimed at reducing and, finally, omitting the amount of FBS in cell culture. The group around Prof Fischer could successfully adapt nine commonly used cell lines to chemically defined (meaning that all components are known), serum-free medium (media such as TurboDoma, Hektor, and In Vitrus from Cell Culture Technologies were used). The benefits of chemically-defined media are clear: all ingredients of the medium are precisely known, which reduces the variability in culturing of cells and bears advantages in down-stream processing, e.g. when cell culture products need to be purified. However, the proliferation rate in chemically defined medium was slightly reduced and in some cases the adaptation caused changes in the phenotype of the cell compared to the controls that were maintained in medium containing FBS (González Hernández and Fischer, 2006). Data like these underline that serum-free cell culture might still be in its infancy, but progress is constantly made. An overview about the current serum-free cell lines and media can be found in a free of charge database supported by the 3R Research Foundation Switzerland. Furthermore, serum-free cell lines are commercially available at the European Collection of Authenticated Cell Cultures (ECACC), where e.g. a serum-free CHO cell line can be purchased. This cell line can be cultured in protein-free (and, thus, serum-free) medium.
Recycling – also popular in science
It is known that serum, rather than plasma, promotes the growth and proliferation of cultured cells (Gospodarowicz and Ill, 1980). Scientists have tried to analyse the composition of FBS for decades and one important finding was that many mitogenic factors present in serum are derived from thrombocytes (Balk et al., 1981; Gospodarowicz and Ill, 1980). This finding suggested that platelet lysates might be a good substitute for animal-derived serum. Activated platelets are known to release a broad spectrum of growth factors and other active molecules by exocytosis (Kind and Reed, 2002). In 2005 and 2011, two studies were published showing that platelet lysates are very efficient in terms of cell proliferation with regard to mesenchymal stem cells and MDCK cells, where 5% platelet lysate had almost the same growth-promoting effect as 10% FBS (Doucet et al., 2005, Rauch et al., 2011). In the latter, outdated human donor platelets were provided by the blood bank of the Innsbruck Medical University Hospital. Donor platelets can be used for five days for therapeutic applications, but it is known that they retain their growth factors for at least three weeks (Chan et al., 2005). Thus, human platelet lysates bear a high potential as FBS substitute in cell culture, since they showed growth promoting effects in a multitude of cell lines (Rauch et al., 2011). However, if the time-sensitive distribution and varying availability of outdated blood samples is applicable for daily cell culture routine remains to be examined.
Our conclusion:
The materials and methods stated above are only some hand-picked examples for the successful replacement of animal-based products in (basic) research. Techniques like those might not be applicable in every lab, but it is definitely worth to think about and be aware of alternatives. If you have worked with one of the methods mentioned in this article or know of further examples for animal-free science, do not hesitate to contact us! Science is about expanding frontiers and maybe one day the BMEL prize money is awarded to a lab based in Bonn. 😉
Literature
Russell, W.M.S. and Burch, R.L., 1959. The Principles of Humane Experimental Technique, Methuen, London
Hodgson, J., 1995. To treat or not to treat: that is the question for serum. Bio/Technology 13, 333–334. 337-338, 342-333.
Jochems, C.E., van der Valk, J.B., Stafleu, F.R., Baumans, V., 2002. The use of fetal bovine serum: ethical or scientific problem? Alternatives To Laboratory Animals 30, 219–227.
Mellor, D.J., Gregory, N.G., 2003. Responsiveness, behavioural arousal and awareness in fetal and newborn lambs: experimental, practical and therapeutic implications. New Zealand Veterinary Journal 51, 2–13.
Van der Valk, J., Mellor, D., Brands, R., Fischer, R., Gruber, F., Gstraunthaler, G., Hellebrekers, L., Hyllner, J., Jonker, F.H., Prieto, P., Thalen, M., Baumans, V., 2004. The humane collection of fetal bovine serum and possibilities for serum-free cell and tissue culture. Toxicology In Vitro, 18(1):1-12.
González Hernández, Y. and Fischer, R. W., 2006. Serum-free Culturing of Mammalian Cells – Adaptation to and Cryopreservation in Fully Defined Media. ALTEX, 24(2):110-6.
Gospodarowicz, D. and Ill, C.R., 1980. Do plasma and serum have different abilities to promote cell growth? Proceedings of the National Academy of Sciences of the United States of America, 77(5):2726-30.
Balk, S.D., Levine, S.P., Young, L.L., LaFleur, M.M., Raymond, N.M., 1981. Mitogenic factors present in serum but not in plasma. Proceedings of the National Academy of Sciences of the United States of America, 78(9):5656-60.
King, S.M. and Reed, G.L., 2002. Development of platelet secretory granules. Seminars in Cell and Developmental Biology, 13(4):293-302.
Doucet, C., Ernou, I., Zhang, Y., Llense, J.R., Begot, L., Holy, X., Lataillade, J.J., 2005. Platelet lysates promote mesenchymal stem cell expansion: a safety substitute for animal serum in cell-based therapy applications. Journal of Cell Physiology, 205(2):228-36.
Rauch, C., Feifel, E., Amann, E.M., Spötl, H.P., Schennach, H., Pfaller, W., Gstraunthaler, G., 2011. Alternatives to the use of fetal bovine serum: human platelet lysates as a serum substitute in cell culture media. ALTEX, 28(4):305-16.
Chan, R.K., Liu, P., Lew, D.H., Ibrahim, S.I., Srey, R., Valeri, C.R., Hechtman, H.B., Orgill, D.P., 2005. Expired liquid preserved platelet releasates retain proliferative activity. Journal of Surgical Research, 126(1):55-8.

Author: Sophie Schonauer

According to Value Market Research, the latest technology trends and global market opportunity analysis in the Cell Culture Media Market industry growing with a high CAGR in the upcoming year. Our report has categorized the market based on technology, service, development, vertical and region.