“We can begin to dare dreaming about curing melanoma”,
a sentence I’ve heard said out loud on the podium during last year’s Society of Melanoma Research (SMR) conference in Boston, the largest and most important conference for all those involved in the fight against malignant melanoma. This sentence would not have been thinkable only a few years ago. Back then, classical chemotherapy using the DNA alkylating drug Dacarbazine was the only available treatment for advanced melanoma. Starting from 2002 the finding of the BRAFV600E mutation hyperactivating the MAPK signaling cascade and driving about 50% of melanomas finally led to the introduction of targeted therapy using the BRAF specific inhibitors Dabrafenib and Vemurafenib. However, despite the initial stunning clinical responses, patients relapse after a median of 5-8 months.
So, what has changed the prognosis for some melanoma patients so drastically in such a short time? The answer seems quite feasible in the retrospective: It is the reactivation of the patients’ own immune system to identify and eliminate malignant cells. However, the road has been a long one to travel and the destination to reach is only about to become visible.
With this blog post I want to share with you the thrilling story of melanoma immunotherapy and how I encountered it during the journey of my PhD thesis.
A long road to travel
Although the “Cancer Immunosurveillance” hypothesis, claiming a reciprocal interaction of cancer and immune cells, was already drafted by Burnet and Thomas back in 1957 (1), there was tremendous doubt due to a lack of experimental evidence for a very long time. It took almost 50 years until Schreiber and colleagues finally presented their refined concept of “Cancer Immunoediting”: During the Elimination phase, malignantly transformed cells are recognized by the immune system on basis of abnormal protein expression and processing. In the consequence, they are destroyed either by direct recognition of immunogenic epitopes or indirectly by activating innate defense mechanisms. However, tumors exhibit tremendous genetic instability, heterogeneity, and plasticity – features that endow them to co-evolve with their immune cell microenvironment. This means that there is constant selection pressure on tumor cells that can hide from immune destruction. At a phase of Equilibrium, there are about as many malignant cells destroyed by the ongoing anti-cancer immune response as malignant cells surviving the immune attack due to the successive acquisition of immunosuppressive traits. In the final phase of Escape, malignantly transformed cells have acquired enough features to completely evade immune cell recognition and will outgrow and manifest as cancer in patients (2).
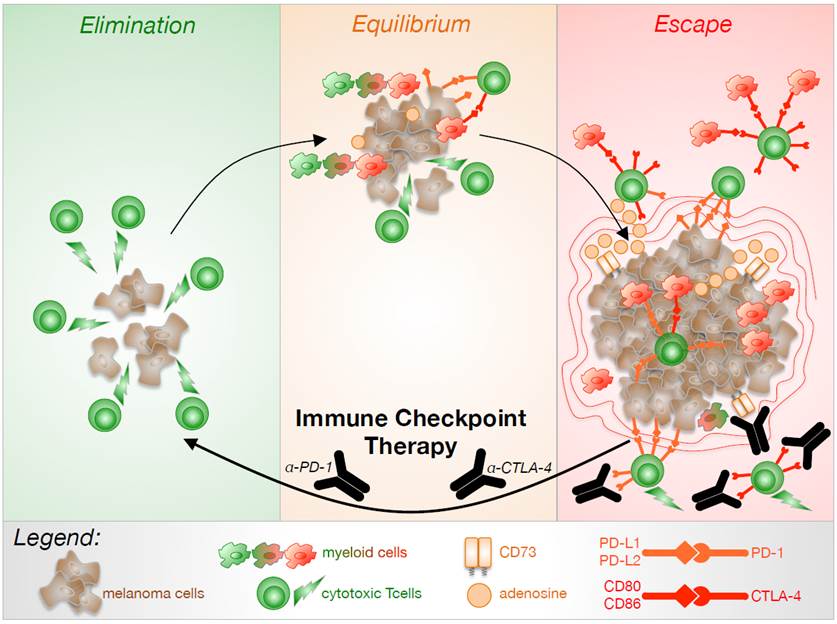
(by J. Reinhardt)
The concept of “Cancer Immunoediting“ was strengthened by more and more research supporting the notion that tumor-immune cell interactions control cancer development and progression. The idea of treating cancer by supporting the patient’s own anti-cancer immune response finally led to the birth of cancer immunotherapy.
My journey to tumor immunology
My first lab encounter with tumor immunology dates back to my undergraduate studies when I worked on a project characterizing tumor infiltrating lymphocytes (TILs) and NK cells in colorectal cancer biopsies in the lab of Prof. Philipp Beckhove at the DKFZ. I stayed on the same road for my Diploma thesis and focused on microglia, the brain’s macrophages, and their potential to phagocytose glioma cells. For my PhD thesis, I joined Prof. Michael Hölzel’s lab and started to investigate how melanoma cells regulate the expression of the immunosuppressive surface-enzyme CD73, a molecule which might serve as a future target in melanoma immunotherapy. CD73 is a 5’ ectonucleotidase expressed by both, tumor and a variety of immune cells, that converts adenosine monophosphate (AMP) to adenosine, thereby promoting an anti-inflammatory microenvironment. Adenosine shuts down anti-tumor immune responses by reprogramming myeloid immune cells and inhibiting T cell function. Recently, we found that CD73 in melanoma is regulated by MAPK signaling involving the transcription factor cJUN (3).
On the SMR conference in Zürich 2014, I witnessed the presentation of Georgina Long who said that about 73% of advanced melanoma patients survive after one year of Nivolumab treatment; a thrilling result when compared to only 42% of patients being still alive in the chemotherapy control group (4). Today, Nivolumab and Pembrolizumab are clinically approved antibodies targeting the negative immunomodulatory receptor programmed death 1 (PD-1) on T and B cells. PD-1 bind to PD-L1 and PD-L2 is induced in the course of inflammation to prevent excess damage, an axis frequently harnessed by cancer cells to prevent anti-tumor immune responses. Another so-called immune checkpoint that negatively regulates T cell activation in the early phase is the high affinity molecule CTLA-4 that outcompetes CD28 to prevent co-activating signals during T cell licensing. The journey of the immune checkpoint blockade began in 2009 by targeting this molecule using the antibody Ipilumumab. For the first time, clinical results clearly superior to the standard chemotherapeutic treatment for advanced melanoma were reported, and researchers started to become excited about the many opportunities that immunotherapy might offer to successfully fight cancer (read more). To appreciate the excitement resulting from the approach of immune checkpoint therapy one has to keep in mind that the road to successful immunotherapy has led through a long desert path to finally hit some fresh water: Many attempts had already been made to reactivate an anti-tumor immune response, among them adoptive T cell transfer, dendritic cell vaccination, and high-dose IL-2 treatment, but they did not yield comparable success rates.
Even though immune checkpoint therapy significantly prolongs melanoma patients’ lives, there is still one major drawback: Only a minority of patients responds to the treatment at all. Therefore, there is an urgent need to understand the susceptibility to immunotherapy, to define novel immunomodulators to be targeted, and to develop combinatorial approaches with acceptable adverse effects (read more).
Quite nervously preparing my talk at the SMR 2016
I felt very much honored when I was invited to orally present my research at the SMR 2016 conference, which was the same conference I had witnessed the fascinating talk by Georgina Long two years ago. And I have probably never felt more nervous and excited at the same time because it felt like if I was allowed to make a tiny little contribution in that huge fascinating story, and all the long exhausting hours in the lab might have been worth it.
To be honest, there were many moments during preparation of my talk when I wished I had not been selected to present, and I was shaking especially the days before the meeting. Surprisingly, I was really calm as soon as I started talking on stage in Boston, realizing that people were interested and positive about what I was going to present. In the end, it turned out to be the right decision to overcome my previous fears and to just join and enjoy the science presented and discussed. And again, there was that hopeful spirit of basic research potentially revolutionizing clinical treatments that I had felt on that previous interdisciplinary conference.
I am very happy to take part in the journey of tumor immunology and I am curious about future developments, as I am sure we’re only starting to understand the mechanisms of tumor immune evasion. To me it still sounds like a dream to fully combat advanced melanoma one day, but obviously “You have to attempt the impossible in order to achieve the possible“ as Hermann Hesse said.
References:
- Burnet M (Apr 1957). “Cancer; a biological approach. I. The processes of control”. British Medical Journal. 1 (5022): 779–86.
- Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (Nov 2002). “Cancer immunoediting: from immunosurveillance to tumor escape”. Nature Immunology. 3 (11): 991–8.
- Reinhardt J et al. (Jun 2017) “MAPK signaling and inflammation link melanoma phenotype switching to induction of CD73 during immunotherapy”. Cancer Res. (Epub ahead of print).
- Robert C et al. (Jan 2015) “Nivolumab in previously untreated melanoma without BRAF mutation”. NEJM. 372(4):320-30.
Julia Reinhardt

Pingback: Interested in Immune regulation, big data modelling, and networking? | ImmunosensationBlog %