In Chinese life philosophy, everything in nature consist of two contradictory states, called, Yin-yang, which are also interdependent, therefore, might be seen as the law of the “unity of the opposite”. Additionally, in traditional Chinese medicine, the yin-yang philosophy acts as a strong foundation for health (Ying) and disease (Yang) as well as treatment approaches. Ying-yang philosophy could also be incorporated into the pathophysiology of sepsis. Sepsis is one of the leading causes of mortality in hospitalized patients worldwide. Clinically it is defined as a potentially fatal organ dysfunction caused by the dysregulated immune response to infection [1]. In the classical sepsis article written by Roger Bone [2], he compares Isaac Newton’s law of “Action” and “Reaction” with the “hyper-inflammation” and “immunosuppression” phases of sepsis. The Ying-yang philosophy could be incorporated into that Bone´s analogy. We can replace the term “hyper-inflammation” by “Yin” and immunosuppression by “Yang” in a perfect way. Today we know that both sepsis states (Yin-yang) can occur not only in sequence to one another but also side by side according to the “unity of the opposite”.
New “Yin” infant sepsis player
One of the best examples of Yin-yang philosophy is INFANT SEPSIS. The hallmark of the infant Yin sepsis phase (hyper-inflammation phase) is the overwhelming organ damage leading to a higher risk of death than adult septic patients [3]. Recently, we observed that this striking organ damage relies on the shocking production of a kind of traps, structure now known as NETs (Neutrophils Extracellular Traps). These are networks of extracellular fibers, primarily composed of DNA from neutrophils associated with proteins (imagine a spider net!) that rapidly trap and kill microorganism.
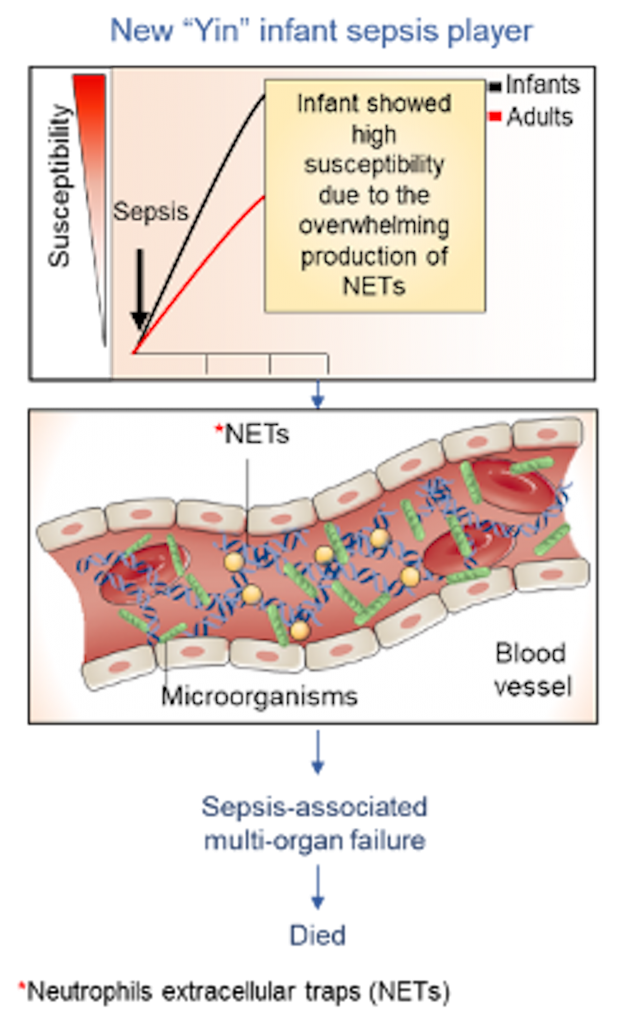
Nevertheless, NETs are a double-edged sword, because they not only kill invading microorganisms but also our cells. In the context of sepsis, we found that infant sepsis individuals produced 40% more NETs than adult counterparts. Strikingly, the serum levels of NETs were positively correlated with the risk of death of infant patients by sepsis. This new Yin infant sepsis player, revealing a hitherto unrecognized mechanism of pediatric sepsis susceptibility and suggest that NETs represent a potential target to improve clinical outcomes of sepsis [5].
Sepsis “Yang”: consequences of sepsis
If you think that just controlling the NETs and the cytokine storm in the acute phase of sepsis is enough to save sepsis patients, rethink that! The problem with sepsis is not only the acute severity and mortality but its consequences, in other words, after sepsis you are not the same, your immune response changes! In the context of adults, the vast majority sepsis-surviving patients can prompt an event of “immunoparalysis” which increases their likelihood of developing secondary infections with opportunistic bacteria or fungi and unfortunately, they succumb at later time points [6-9]. Numerous studies have been demonstrated that this Yang sepsis phase or “sepsis long-term immunosuppression” might last for up to five years! [10]. Recently, we demonstrated that this condition is due to the overwhelming expansion of different subset of anti-inflammatory cell types such as regulatory T cells or Tregs and anti-inflammatory macrophages called “M2 macrophages” as well as, not less important, the high production of IL-33 (soluble mediator released by our cells during injury) [11-13]. Likewise, our group recently demonstrated in mice and human settings that IL-33 is the “master regulator” for the development of post-septic immunosuppression [13].
Unexpected “Yang” in infant sepsis survivors
The examination of the long-term immune consequences of sepsis in the pediatric population has been neglected. Remarkably, in a set of experiments through the infection of sepsis-surviving animals with bacteria or using a tumor model, sepsis-surviving infant mice were totally resistant to bacteria secondary infection as well as did not show changes in tumor control when compared to adults. Our findings suggest that despite the high mortality of infant mice in the acute phase of sepsis (Yin phase), if they survive to sepsis, sepsis-surviving infant mice do not develop late immunosuppression, unlike previously reported in adult individuals, this is an “unexpected Yang”. Following our findings, we explore the production of the immunosuppression master regulator, IL-33, as well as the suppressor cell populations (regulatory T cells and M2 macrophages) in sepsis-surviving infant mice. Surprisingly, infant survivors showed a marked reduction of these anti-inflammatory mediators. Taking these findings into account, we decided to move to human settings and we demonstrated that similar to shown in mice sepsis model, sepsis-surviving pediatric patients did not show Tregs cell expansion or increase in serum IL-33 levels when compared to adults.
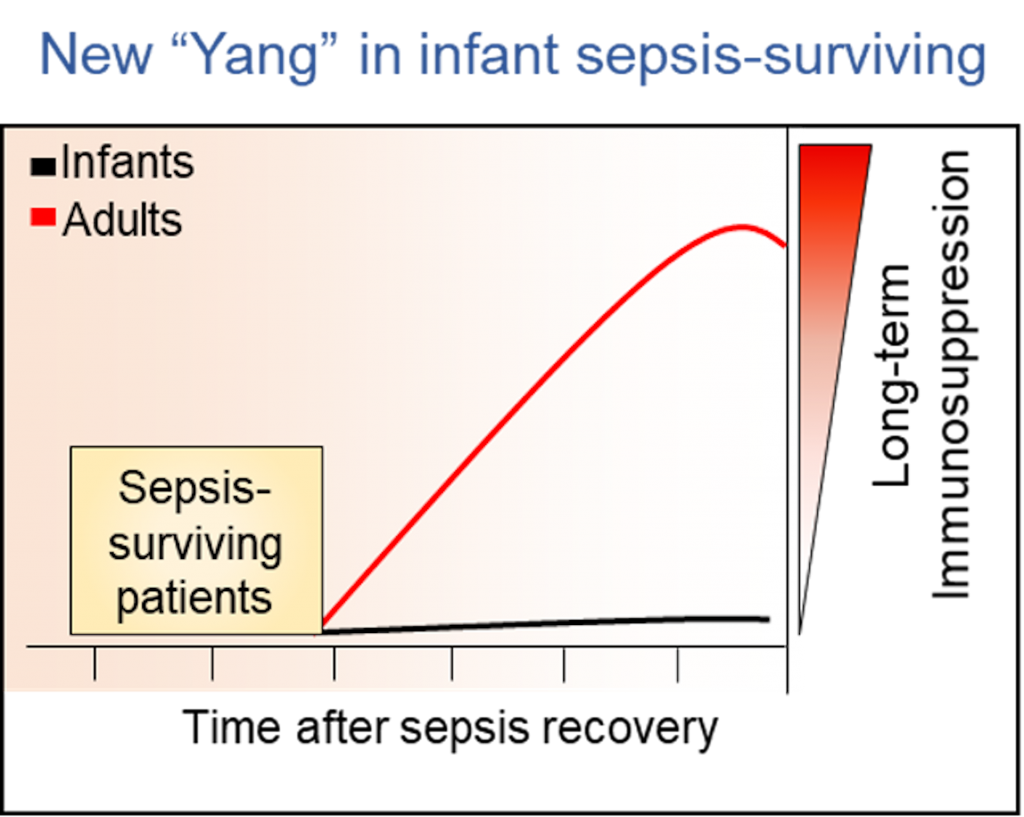
To conclude, we have shown that, even though in the acute phase of sepsis (Ying phase), infants are highly susceptible; after the septic process (Yang phase), sepsis-surviving mice are “resistant” to the development of long-term immune dysfunction, suggesting that long-term immunosuppression after sepsis is an unappreciated age-dependent phenomenon. Therefore, infant sepsis is a perfect example of Yin-yang philosophy: “UNITY OF THE OPPOSITE”.

Written by David Fernando Colón Morelo
References
1. Singer, M., et al., The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA, 2016. 315(8): p. 801-10.
2. Bone, R.C., Sir Isaac Newton, sepsis, SIRS, and CARS. Crit Care Med, 1996. 24(7): p. 1125-8.
3. Zhao, J., et al., Hyper innate responses in neonates lead to increased morbidity and mortality after infection. Proc Natl Acad Sci U S A, 2008. 105(21): p. 7528-33.
4. Papayannopoulos, V., Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol, 2018. 18(2): p. 134-147.
5. Colon, D.F., et al., Neutrophil extracellular traps (NETs) exacerbate severity of infant sepsis. Crit Care, 2019. 23(1): p. 113.
6. Meakins, J.L., et al., Delayed hypersensitivity: indicator of acquired failure of host defenses in sepsis and trauma. Ann Surg, 1977. 186(3): p. 241-50.
7. Wang, T., et al., Subsequent infections in survivors of sepsis: epidemiology and outcomes. J Intensive Care Med, 2014. 29(2): p. 87-95.
8. Hotchkiss, R.S., G. Monneret, and D. Payen, Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol, 2013. 13(12): p. 862-74.
9. Otto, G.P., et al., The late phase of sepsis is characterized by an increased microbiological burden and death rate. Crit Care, 2011. 15(4): p. R183.
10. Muenzer, J.T., et al., Characterization and modulation of the immunosuppressive phase of sepsis. Infect Immun, 2010. 78(4): p. 1582-92.
11. Nascimento, D.C., et al., Role of regulatory T cells in long-term immune dysfunction associated with severe sepsis. Crit Care Med, 2010. 38(8): p. 1718-25.
12. Cavassani, K.A., et al., The post sepsis-induced expansion and enhanced function of regulatory T cells create an environment to potentiate tumor growth. Blood, 2010. 115(22): p. 4403-11.
13. Nascimento, D.C., et al., IL-33 contributes to sepsis-induced long-term immunosuppression by expanding the regulatory T cell population. Nat Commun, 2017. 8: p. 14919.
