Back pain, cravings, weight gain, and hormonal changes are just a few of the many (wonderful) things that happen during pregnancy. During this period of life, the immune system of the pregnant parent similarly undergoes many changes. Some decades ago, scientists and physicians thought that one of these changes included the suppression of the immune system. Therefore, it was believed that a rejection of the baby by the body of the mother was avoided. Based on this hypothesis, a pregnant mother was long thought to be prone to infections and diseases. We now know that the immunology of pregnancy is way more complex than the mere suppression of the immune system, and that it requires fine-tuning of several immunological processes.

For a successful pregnancy, pro-inflammatory and anti-inflammatory processes must occur at the right time and place. A pro-inflammatory response means that the inflammation in the area is encouraged, and as a result, blood vessels become leakier and many immune cells are recruited. An anti-inflammatory response, on the contrary, reduces inflammation, supports healing, and gives the area some tolerance to the stimulus that caused the inflammation. For example, a bee sting can drive a pro-inflammatory response, which you could afterward alleviate by taking an anti-inflammatory drug like ibuprofen.
Immunity in pregnancy: Early stage
The blastocyst is the name biologists give to a fertilized egg cell that has already undergone some cell divisions. It is a bunch of cells that consist of an inner part, which gives rise to the embryo, and an outer part called the trophoblast, which protects it. The blastocyst travels from the fallopian tube to the uterus. Here, it undergoes the process of placentation, in which the blastocyst attaches to the endometrium, and the placenta develops.
In early pregnancy, a fascinating, mutual communication happens between the trophoblast and the lining of the uterus. The trophoblast releases factors to promote the recruitment, maturation, and differentiation of the immune cells in the uterus. At the same time, the surface of the uterus itself changes to promote its adherence which is also called implantation [1].
Image created with Biorender.com
In the implantation process, a local pro-inflammatory response is triggered that allows the fertilized egg to be attached. A pro-inflammatory reaction means that the inflammation in this area is endorsed. As a result, the cells on the surface of the uterus, the so-called epithelial cells, express molecules that help the attachment of the blastocyst. At the same time, the uterine mucus proteins are reduced. The mucus proteins are a viscous and gel-forming secretion that usually protects the uterus from infections. However, they are diminished in this period to facilitate the receptivity of the egg. Not only do the epithelial cells play a role in this process: the dendritic cells underneath provide growth factors that encourage the generation and maturation of blood capillaries and modify the tissue for preparing this area to uphold a developing embryo. In a non-pregnant uterus, there are always some dendritic cells present, but in the early pregnancy, they are gathered, and their numbers steadily increase with the progression of pregnancy [2].
Immunity in pregnancy: Late stages
After the implantation, around the second trimester of the pregnancy, the principal local immune response is anti-inflammatory. This means that the local responses switched from one which promoted inflammation to one that suppressed it. As a part of this suppressing response, many immune cells such as regulatory T-cells (Treg cells), natural killer cells, and macrophages contribute to establishing this environment. Macrophages, the specialized ingesting cells of the immune system, produce anti-inflammatory cytokines and clear dying cells of the trophoblast to prevent the contact of the molecules from the embryo that could cause a reaction by the mother.
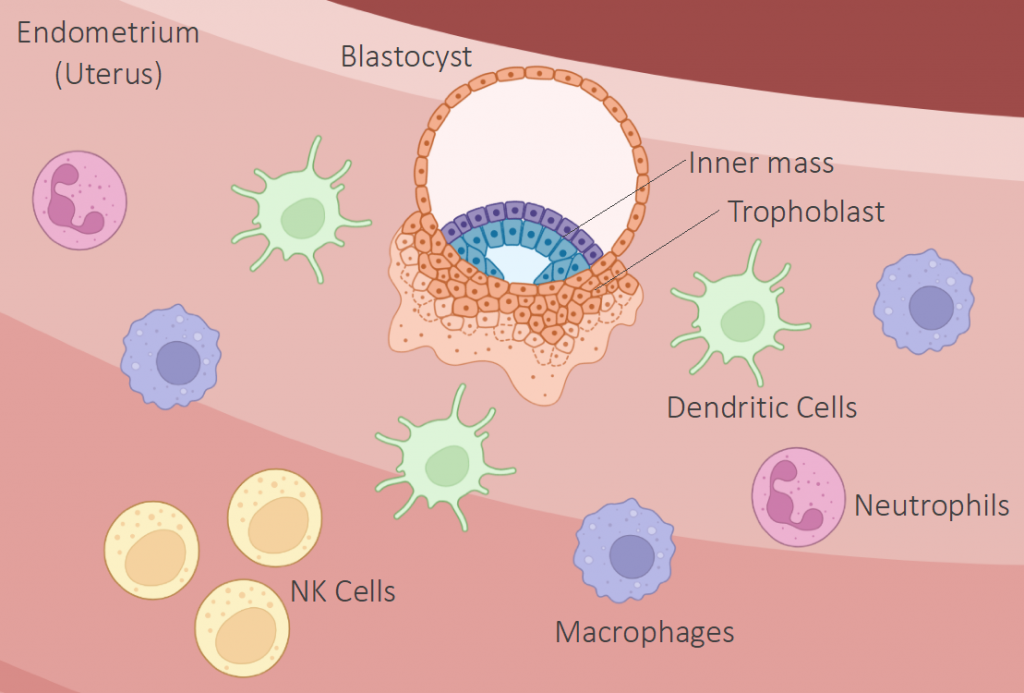
Image created with Biorender.com
Here also, Treg cells and dendritic cells help the uterus be less sensitive to the embryo to avoid a reaction that could result in miscarriage. Treg cells are a type of cells that suppress the immune response. These cells contribute to the protection of molecules in the embryo/fetus that are derived from the father from the immune responses of the mother. Simultaneously, dendritic cells profile the molecules that help the maternal environment tolerate the fetus.
When the fetus is developed, a switchback into a pro-inflammatory response to support delivery happens. At this point, molecules produced by the developing fetus trigger the contractility of the uterus. For example, proteins from the baby’s lung activate the so-called Toll-like receptors 4 (TLR4) present in the cells of the uterus [3,4]. This activation promotes a series of responses, including the gathering of immune cells in the myometrium, where the muscle cells of the uterus are found. As a result, the myometrium dilates its blood vessels, which prepares and supports the contractions necessary to give birth naturally to a new human being.
What happens if there is no tuning of the inflammatory responses?
On one side, local and different inflammation responses are crucial for a successful pregnancy. On the other, general, chronic inflammation of the maternal body can result in adverse outcomes for the offspring. This is called in the scientific jargon “maternal immune activation” and can be triggered by different factors from a viral infection, stress, obesity to an exposure to drugs. The outcomes for the fetus are often preterm birth or even miscarriage, and even consequences for its health like susceptibilities to infections, allergies, or even learning disabilities [5,6].
Some last words
Pregnancy is not characterized by strong immune suppression. Interestingly, many researchers even compare the immunology of pregnancy rather with tumor responses [1]. As controversial as this might sound, cancer cells promote tolerance through gradual antigen presentation, endorse the formation of blood vessels, and recruits anti-inflammatory cells after the attachment to encourage its growth, which is pretty similar to the cross-talk between the uterine dendritic cells and blastocyst, and like the increased tolerance to the fetus mediated by macrophages, Treg cells and dendritic cells in the second trimester of pregnancy.
A significant part of the human population will be pregnant at least once in their life. Moreover, every single human on the earth originates from a successful pregnancy (at least until we learn the secrets from Aldous Huxley’s Brave New World). For this reason, we must get a better understanding of this immunological, personal and emotional state.
Eliana Franco Taveras
Twitter: @AnaileFranco or LinkedIn: rb.gy/beeayu
Bibliography and recommendations
- Mor, G., Aldo, P. & Alvero, A. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol 17, 469–482 (2017). https://doi.org/10.1038/nri.2017.64
- Plaks V, Birnberg T, Berkutzki T, et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J Clin Invest. 2008;118(12):3954-3965. doi:10.1172/JCI36682
- Lindström, Tamsin M, and Phillip R Bennett. “The role of nuclear factor kappa B in human labor.” Reproduction (Cambridge, England) vol. 130,5 (2005): 569-81. doi:10.1530/rep.1.00197
- Behnia, F., Sheller, S. & Menon, R. “Mechanistic differences leading to infectious and sterile inflammation.” Am. J. Reprod. Immunol. 75, 505–518 (2016).
- Minakova, E, Warner, BB. Maternal immune activation, central nervous system development, and behavioral phenotypes. Birth Defects Research. (2018); 110: 1539– 1550. https://doi.org/10.1002/bdr2.1416
- Thornton, Catherine A., Trisha V. MacFarlane, and Patrick G. Holt. (2010). “The Hygiene Hypothesis Revisited: Role of Materno-Fetal Interactions.” Current Allergy and Asthma Reports. https://doi.org/10.1007/s11882-010-0148-5.
… And inspired by the presentation by Prof. Dr. med. Prazeres da Costa in the Autumn School of the DGfI’s Academy of Immunology.
If you would like to know more about maternal immune activation, check out the work by the labs of Prof. Dr. med. Clarissa Prazeres da Costa and Prof. Dr. Elvira Mass!
