The bloodstream is the red carpet for the immune system. Through every capillary, thousands of leukocytes parade and circulate as main actors of innate and adaptive immunity. They come from the bone marrow, dressing their best membrane markers and receptors, waiting for the opportunity of showing their talent on stage, fighting invasions and keeping the homeostasis of the organism. Neutrophils, monocytes and lymphocytes are easily found on the cover of famous journals, in different colors and resolutions, since they are indeed critical cells in the immune response.
However, there is another cell type often forgotten as part of these mechanisms. The platelets have been renowned so far for the performance which brought them to stardom: in healthy conditions, they are responsible for coagulation and bleeding contention, but they can also play the villain as part of the formation of blood clots, leading to conditions such as thrombosis and atherosclerosis. My name is Lucas Secchim Ribeiro and I am a post-doctoral researcher in the AG Franklin, Institute of Innate Immunity. Our research group works to explore the potential of the platelets beyond the hemostasis, introducing their skills as part of our immune system, in health and disease.
Platelets: beyond the script
A single drop of blood holds around 10 million of platelets, second in number only to red blood cells. They are organized pieces of the cytoplasm derived of megakaryocytes coming from the bone marrow and the lungs. However, even without a nucleus, they carry organelles like mitochondria and ribosomes, as well as different types of granules containing nucleic acids, bioactive lipids, proteins and other small molecules that can be released in the form of microvesicles upon activation.
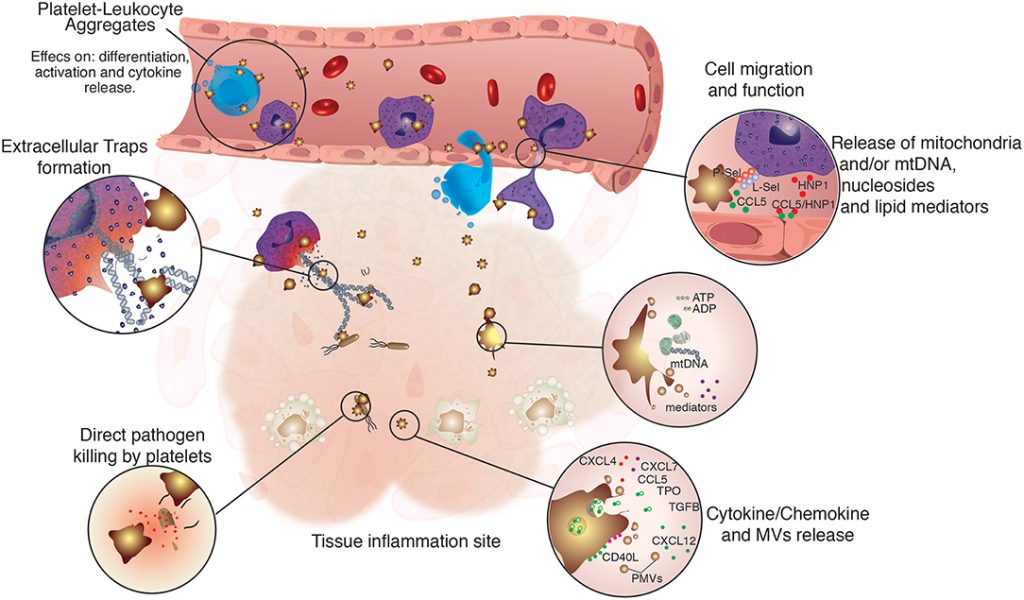
Like their leukocyte counterparts, platelets also express in their membranes a vast collection of receptors able to detect molecular patterns from pathogens or internal signs of danger (PAMPs and DAMPs, respectively). By using these receptors, platelets recognize invading microbes or infected cells and send signals to other immune cells, in order to provide the purge of the inflammatory agent. Besides, platelets can sense the injury and express in their own membrane a type of homing beacon known as selectin, guiding leukocytes to the source of the problem and leading to the elimination of the threat. In a specific interaction with neutrophils, for instance, platelets serve as a scaffold for the formation of superstructures known as NETs – neutrophil extracellular traps – whose goal is to increase the area of capture and extermination of circulating bacteria.
Moreover, platelets are not contained to the blood vessels: they are equally able to infiltrate an infected tissue and initiate the restraint of the pathogen, further signaling and increasing the killing activity of phagocytes. This augmented immune activity can also be perceived by observing the formation of platelet-leukocyte aggregates: by using molecular anchors, both cell types circulate with higher potential for inflammatory activity, dealing with bacteria and parasites with utmost efficiency.
Inflammasomes, caspase, action!
Leukocytes are fundamental for the beginning and resolution of the inflammatory process. An important component of this system is the inflammasome – a complex set of proteins that is assembled to promote the activation of enforcer enzymes named caspases. Once active, the caspases will lead to the maturation of inflammatory cytokines, like interleukin-1 beta and interleukin-18. Finally, a protein named gasdermin D will drive the formation of pores in the membrane, with the leaking of cytoplasmic material and sealing the deadly fate of the cell.
Given its deleterious capability, the activation of the inflammasome is tightly regulated to maintain the body in perfect activity or homeostasis. However, this control can be broken due to genetic conditions and in that case, the organism can overreact and cause damage to itself. For instance, Muckle–Wells syndrome, Neonatal-onset Multisystem Inflammatory Disorder, gout, Alzheimer´s disease and rheumatoid arthritis are conditions associated with activating mutations on NLRP3, the best studied inflammasome (nicely introduced in a previous post).
Many studies on inflammasomes dissect cellular mechanisms by showing patterns of response in monocultures of immune cells. However, it is well known that these cells are rarely isolated in the body. Therefore, our research group raised the hypothesis that the interaction between platelets and leukocytes is an important source of modulation for the immune system.
In our experiments, we study the innate immune response of different types of leukocytes, like macrophages and neutrophils, in the presence of platelets. The stimulation of the inflammasomes prompts the release of IL-1b by the leukocytes, but in the co-culture with platelets the production of this cytokine is significantly higher, as well as for IL-18, another inflammasome-derived marker. These findings were confirmed in both human and mouse cells. The boost in the cytokine production is not attributed to an additive effect from the platelets, because we are not able to detect IL-1b or any other inflammasome component in these cells.
Since platelets are known to secrete important mediators from their granules, we tested if the cell-free supernatant from freshly isolated platelets would be able to replicate the effect observed and the result was positive: both platelets or their releasates can equally enhance the activation of the inflammasome in leukocytes, as also shown by the higher activity of caspase-1, the final and fundamental enzyme in the inflammasome assembly. The incremental effect of the platelet supernatants was lost after thermal and chemical inactivation. Based on these results, a deep evaluation of the protein and nucleic acid content of the platelet supernatants is being performed in order to identify the possible candidate(s) responsible for this event.
Corroborating our data, we also found that children with an overreactive mutation in the NLRP3 gene have an increased expression of platelet-related genes when compared to healthy controls. Moreover, we also found a positive correlation between the platelet counts and the plasmatic concentration of IL-1b in malaria patients from an endemic area. Therefore, in conclusion, our results prove the importance of platelets in the regulation of the inflammasome and its downstream effects.
From supporting role to rising star
As we face “The End” and the final curtains of this post, we hope to see brighter days for these amazing cells. For too long, platelets have been solely linked to coagulation and thrombotic conditions but the recent scientific discoveries have raised the awareness of platelets as a main character on the stage of immunology. The small, wandering cell was found entering tissues to fight infections and guiding other cells, attracting the flashes of attention from scientists all around the world. Together with their established function within the circulatory system, further studies of the interaction between platelets and the other cells in the body will help to identify possible therapeutical strategies, expanding our knowledge of their effect in health and disease. The platelets are now on their way to recognition and appreciation as full members of the select group of immune cells. The show must go on!
Author: Lucas Secchim Ribeiro
